 N2 (g) This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Which of the following is true about the numerical value of Gibbs free energy change for a spontaneous reaction. Chemistry questions and answers Which one of the following has the highest standard molar entropy, S, at 25C A. Which system has the greatest entropy 1 mol of H2(g) at 100C 0.5 atm has the largest entropy because it is in gaseous form and it has the highest temperature. Derivation: The Helmholtz equation is derived using the. How many joules are needed to warm 5.35g from 35 C to 43 C. T Absolute temperature of the surroundings in Kelvin S Entropy of the system in joules per Kelvin. The specific heat of silver is 0.24 J / g C. If you don’t remember the order followed by the entropy in states of matter, just identify the gaseous molecule with the lower molecular mass. Which one of the following systems has the highest entropy. Note: Don’t get confused while determining the molecule with the higher entropy. Thus, in the last we can conclude that the one molecule of hydrogen gas has the highest entropy. (4) The dissolving of the LiBr(s) in water is an exothermic process. (3) The dissolving of the LiBr(s) in water is an endothermic process. The third law of thermodynamics establishes the zero for entropy as that of a perfect, pure crystalline solid at 0 K. If Suniv < 0, the process is nonspontaneous, and if Suniv 0, the system is at equilibrium. It means the substance with the less molar mass will have higher entropy. (2) The entropy of the LiBr(aq) is less than the entropy of the water. The second law of thermodynamics states that a spontaneous process increases the entropy of the universe, Suniv > 0. We can also measure the entropy in consideration with the molar mass. 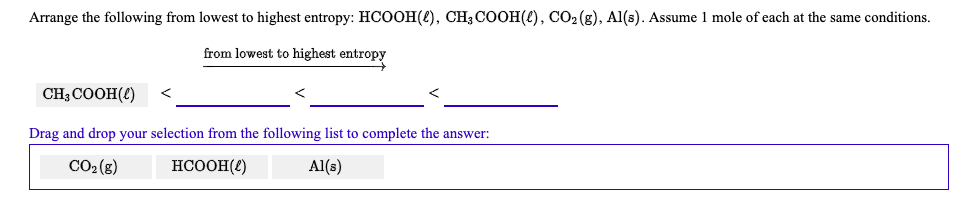 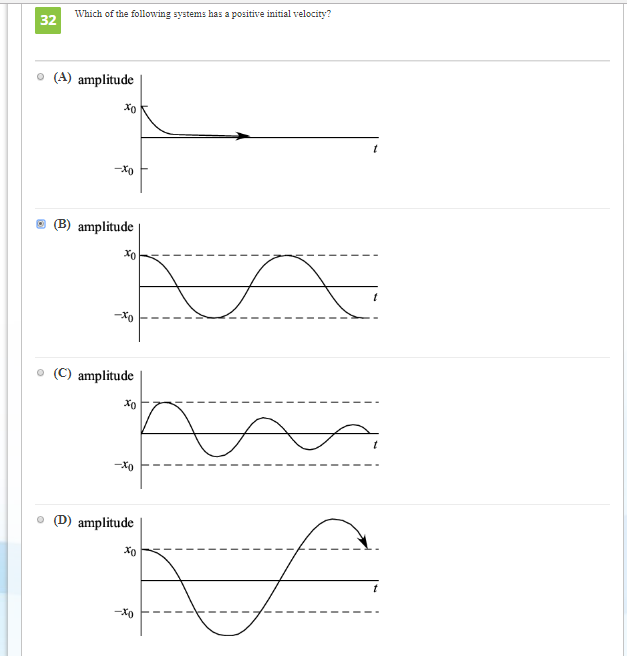
Talking about hydrogen gas, it exists in the gaseous state. > Now, the next if we consider mercury, and liquid nitrogen we know that these both molecules exist in the liquid state. So, if we look at the given options, the diamond exists in the solid state, and is an allotrope of carbon. The order followed is gas > liquid > solids > Thus, the greater disorder, and the system having high temperature will lead to the increase in entropy. Which law states 'Energy can neither be created nor. Which law states 'The entropy of a closed system is always increasing' Zeroth Law. > In terms of thermodynamics, it is used to determine the behaviour in terms of temperature, pressure of a system whereas statistical definition tells us that it is used to measure the molecular disorder in a system. Which of the following would have the highest entropy A cold solid. Entropy is defined on the basis of thermodynamics, and statistics. There is an order followed by entropy in the three states of matter, with the help of that, the molecule can be determined. 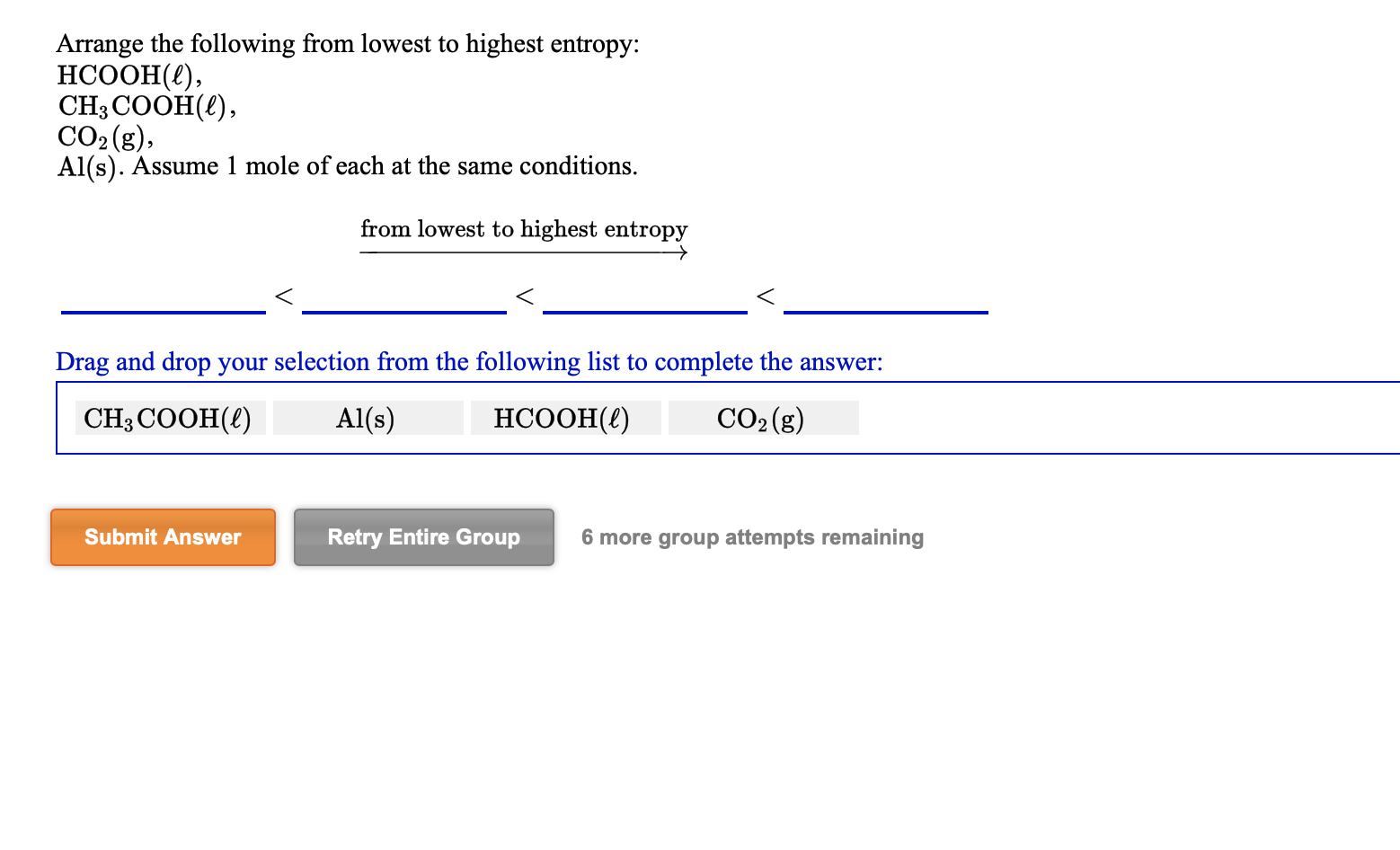
We know that entropy is a measurement of disorder in a system. Determine which of the following has the highest entropy at SATP: Cl2(g) Na(s) NaCl(s) NaCl(aq) BUY. Hint:We have to find the molecule with the highest entropy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
